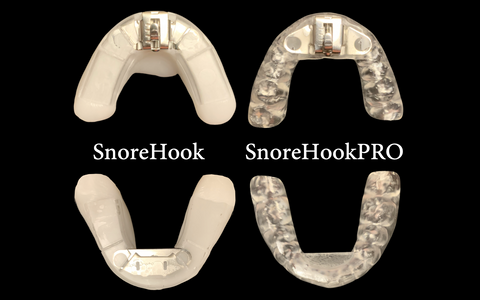
SnoreHook: FDA and PDAC/Medicare approved
CLICK HERE for details about WarpSpeed! SnoreHookPRO same-day fabrication, overnight delivery service.
REQUEST FREE SAMPLE of the chairside fabricated device. See the new SnoreHookPRO
SnoreHook:Morpheus. An upgraded Hooking Mechanism, with cleared billing code HCPCS code K1027
CLICK HERE to download the in-office fabrication protocol
A durable treatment device that is affordable as a trial device!
The SnoreHook Splint patented customizable trays (that can be adapted to any model arch width) combined with the titratable, mandibular-advancing stainless steel hooking mechanism, is FDA cleared for the treatment of snoring and mild to moderate obstructive sleep apnea and as now approved for Medicare insurance code E0486.
- Has a mechanism that is hinged or jointed at the sides, front or palate;
- Has a mechanism that allow the mandible to be advanced;
- Is able to protrude the mandible beyond the front teeth at maximum protrusion;
- Is adjustable by the beneficiary by increments of one millimeter or less;
- Retains its adjustment setting when removed.
The SnoreHook Splint is fabricated on patient's models in the practitioner's in-office lab iat a fraction of the typical lab costs.
Watch this accelerated fabrication video:
(For the realtime fabrication video, with embedded comments and guidance, click here)
Be sure to visit the links in the drop down menu "SnoreHook Help" above for useful insights and procedures, for example, making the "Comfort Backstop" for patient comfort.
Each SnoreHook Splint protocol set includes:
- Mandibular advancing hooking mechanism, screw adjustable in 1mm increments
- Mandibular CrossPlate, which opposes the maxillary hook;
- Pre-engineered patented maxillary and mandibular width-adjustable polycarbonate trays that the Hooking Mechanism and CrossPlate fit into;
- Hexagonal adjusting key, to advance the Advancing Hook;
- Security Bar (to prevent dislodgment during use)
- Ample supply of thermo-acrylic beads to fabricate the devices.


Practitioners: For the best insight on what the patient's experience is, make a SnoreHook on yourself:
How to relieve a tight/binding fit; Lower device insertion/removal
*Lifetime Warranty: If any component of the SnoreHook Splint fails or is damaged, or even if the patient elects not to use the final appliance, return the components and receive replacement components at no charge. (see the FAQs for details).
Practitioners can send their request for a Trial Kit of the SnoreHook Splint to Help.ChairsideSplint@gmail.com. We'll then provide the link to the SnoreHook Trial Kit page and a checkout code for a 100% discount on the kit fee (you'll pay for postage/handling only). We're always interested in how you became aware of the SnoreHook Splint, so please let us know where you've heard or read about it.


